Similar presentations:
Solving the “Tritium Problem” in Nuclear Industry
1.
Solving the “Tritium Problem” in NuclearIndustry.
Sevastopol state university
By Victoria Ulanovskaya
KhT/b-21-1-o
2.
Content:• 1. Introduction
• 2. Some information about tritium, its physical and chemical
properties
• 3. What is the “tritium problem” about?
• 4. Ways to solve the problem
• 5. Conclusion
3.
INTRODUCTIONThe problem of tritium formation in reactors and its
possible release into environment is becoming more and
more relevant. In literature we can find such interesting
term, as “tritium problem”.
The atom of tritium is unique nuclide, which has specific
properties. Because of this scientists and workers of NPP
are interested in this problem.
4.
Some information about tritium, its physical and chemical propertiestritium from anc. Greek τρίτος- “the third”
Tritium is radioactive isotope of hydrogen, the second name
of tritium is triton. In the process of decay, tritium turns into
3He with the emission of an electron and an antineutrino. It
was discovered by Ernest Rutherford, Marcus Oliphant and Paul
Harteck in 1934.
Industrial tritium is obtained by irradiation of lithium-6 with
neutrons in nuclear reactors by the following reaction:
The half-life(T 1\2) of this nuclide is 12.32 years
The available decay energy (E decay) is 18.59 keV
Average energy of electrons is 5,7 keV
The atomic mass is 3.016
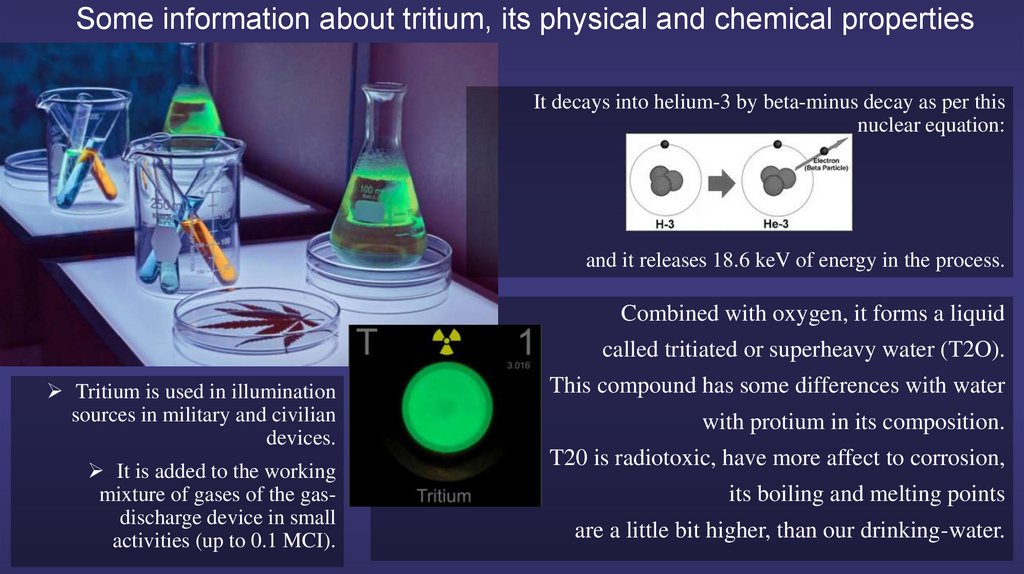
5.
Some information about tritium, its physical and chemical propertiesIt decays into helium-3 by beta-minus decay as per this
nuclear equation:
and it releases 18.6 keV of energy in the process.
Combined with oxygen, it forms a liquid
called tritiated or superheavy water (T2O).
Tritium is used in illumination
sources in military and civilian
devices.
This compound has some differences with water
It is added to the working
mixture of gases of the gasdischarge device in small
activities (up to 0.1 MCI).
T20 is radiotoxic, have more affect to corrosion,
with protium in its composition.
its boiling and melting points
are a little bit higher, than our drinking-water.
6.
What is the “tritium problem” about?The tritium problem is due to the fact that tritium is
synthesized in nuclear reactors (divider, moderator, coolant),
which is released in significant quantities into the
environment.
Solving the problem requires the development of methods
for purifying nuclear waste from tritium, and methods for
isolating tritium and its practical use in different industries.
Tritium is not dangerous when exposed
to the external environment, but when it inhaled,
ingested in food and water,
or absorbed through the skin, it poses a risk
due to internal exposure to radiation.
A significant excess in the amount of
tritium produced in heavy water reactors
is due to the radiation reaction of
neutron capture by deuteron D(n, γ)T
occurring in the moderator and coolant.
Tritium is formed in the reactor core
when uranium and plutonium are
irradiated.
7.
What is the “tritium problem” about?The constant increase in the volume of accumulated
tritium leads to a gradual increase in the amount of tritium
emissions and discharges, which entails an increase in the
impact of ionizing studies on personnel and the population
through the ingestion of tritium water.
To reduce the technogenic impact of tritium on the
environment, the permissible standards for its intake
changed with the development of science and technology.
Bypassing the treatment barriers, the tritium (or
radioactive hydrogen) generated at the nuclear power plant
enters the operating system with liquid discharges in the
form of tritium water and gaseous emissions.
8.
Ways to solve the problemCECE-process(combined electrolysis and catalytic exchange)
Phase isotope exchange
H2O(liq)+ HT(gas) ↔ H2(gas) + HTO(liq)
Complex technology
α = HT/HTO ~ 6.05 ~ 40 °C
Phase isotope exchangе
Integrated technology
It is based on the process of phase isotopic
exchange of water vapor in air with water. This
process is simple and has been mastered on
an industrial scale for absorption purification.
The problem of secondary waste generated during
air purification is solving by following method
This method lies on the fact that natural water
is supplied to the column filled with a nozzle
from above, and a stream of purified air from
below. The concentration of tritium in water
vapor in the air decreases from bottom to top,
and in water it increases. The process is
carried out at a temperature of 20 to 30 ° C.
By changing the height of the column, it is
possible to achieve any required degree of air
purification from tritium.
This method is based on the chemical isotope
exchange of hydrogen with water, and it called
CECE-process . For this process a special catalyst
with the property of hydrophobicity is required. The
facility for deprotization and detritization of
superheavy-water waste has been operating in
Russia since 1995.
9.
Ways to solve the problemWD- process, Water rectification technology
H2O(liq)+ HTO(gas) ↔ H2O(gas)+ HTO(liq)
α = HTO/ HTO ~ 1.056~ 60 °C
Rectification of water under vacuum is one of the most
safe ways to separate hydrogen isotopes.
The method is based on the difference in the degree of
volatility of HTO and H2O. However, this method is
complicated due to the need to use special equipment,
as well as with significant energy costs necessary to
maintain a constant temperature and pressure.
The rectification process is a separation process based
on different boiling points of the components, carried out
by repeated contact of the steam and liquid phases in the
counterflow, as a result of which the steam phase is
enriched with low-boiling components, and the liquid
phase is enriched with high-boiling components.
Columns with poppet and nozzle contact devices are
used to carry out the process of water rectification.
10.
CONCLUSIONBased on the information,which was mentioned below, we
can draw some conclusions about tritium problems. Firstly,
it is a relevant question at the moment, because it concerns
different spheres, such as ecological, industrial, economical,
scientific, and other industries.
Secondly, the methods of detritization aren’t perfect, they’re
have a lot of advantages, but each of them has a several
drawbacks, so scientists try to find more different ways to
solve this problem to minimize the amount of
disadvantages.
But now the methods of purifying water are successfully
used in industrial and scientific purposes.
11.
Thank you for Attention!Sevastopol state university By Victoria Ulanovskaya KhT/b-21-1-o










 ecology
ecology








