Similar presentations:
Сharacteristics of Chemical power sources (CPS)
1. Сharacteristics of Chemical power sources (CPS)
2.
The following battery characteristics must be takeninto consideration when selecting a battery:
Type
Voltage
Charge/Discharge curve
Capacity
Energy density
Temperature dependence
Service life
Self Discharge
3.
1. TypeSee primary and secondary batteries page.
Primary batteries
Secondary batteries
Application
Portable devices
Car battery used to start engine
Lighting
Aircraft systems
Toys
Standby power resources
Memory back-up
Emergency no-fail systems
Watches/clocks
Electric vehicles
Hearing aids
Mobile phones
Radios
Cameras
Medical implants
Power tools
Defence related systems such as missiles Toys
Portable computers
Advantages
High power density
High discharge rate
Good low temperature performance
Inexpensive
Convenient
Lightweight
Good shelf life
High Energy density at low/moderate
discharges
Disadvantages
Can only be used once
Lower Energy density
Leads to large amount of waste batteries Poorer charge retention
to be recycled
Safety issues
Batteries put into landfill sites have
Lack of standards
severe environmental impact
High initial costs
Life cycle energy efficiency < 2 %
4.
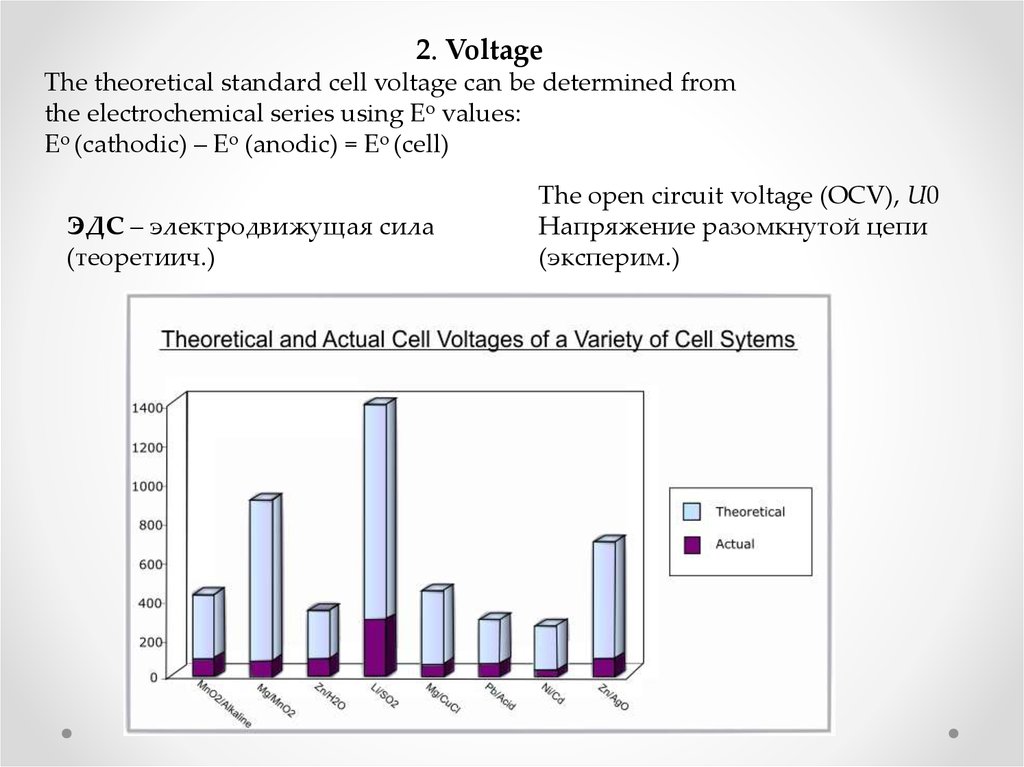
2. VoltageThe theoretical standard cell voltage can be determined from
the electrochemical series using Eo values:
Eo (cathodic) – Eo (anodic) = Eo (cell)
ЭДС – электродвижущая сила
(теоретиич.)
The open circuit voltage (OCV), U0
Напряжение разомкнутой цепи
(эксперим.)
5.
3. The Charge/Discharge CurveThe measured terminal voltage of any battery will vary as it is charged and
discharged (see Figure 1).
The MPV (mid-point voltage) is the nominal voltage of the cell during charge or
discharge. The maximum and minimum voltage excursion from the nominal
value is an important design consideration: a "flatter“ discharge curve means
less voltage variation that the design must tolerate.
When peak charged, the actual cell voltage will be higher than the MPV. When
nearing the EODV (end of discharge voltage) point, the cell voltage will be less
than the MPV. The EODV is sometimes referred to as the EOL (end of life)
voltage by manufacturers.
6.
4. CapacityThe theoretical capacity of a battery is the quantity of electricity involved
in the electro-chemical reaction. It is denoted Q and is given by:
where x = number of moles of reaction, n = number of electrons transferred
per mole of reaction and F = Faraday's constant.
The capacity is usually given in terms of mass, not the number of moles:
where Mr = Molecular Mass. This gives the capacity in units of Amperehours per gram (Ah/g).
Емкость элемента – это количество электричества, которое
химический источник тока отдает при разряде
C = I . (А . ч, если элемент разряжается током I (A) в течение (ч))
7.
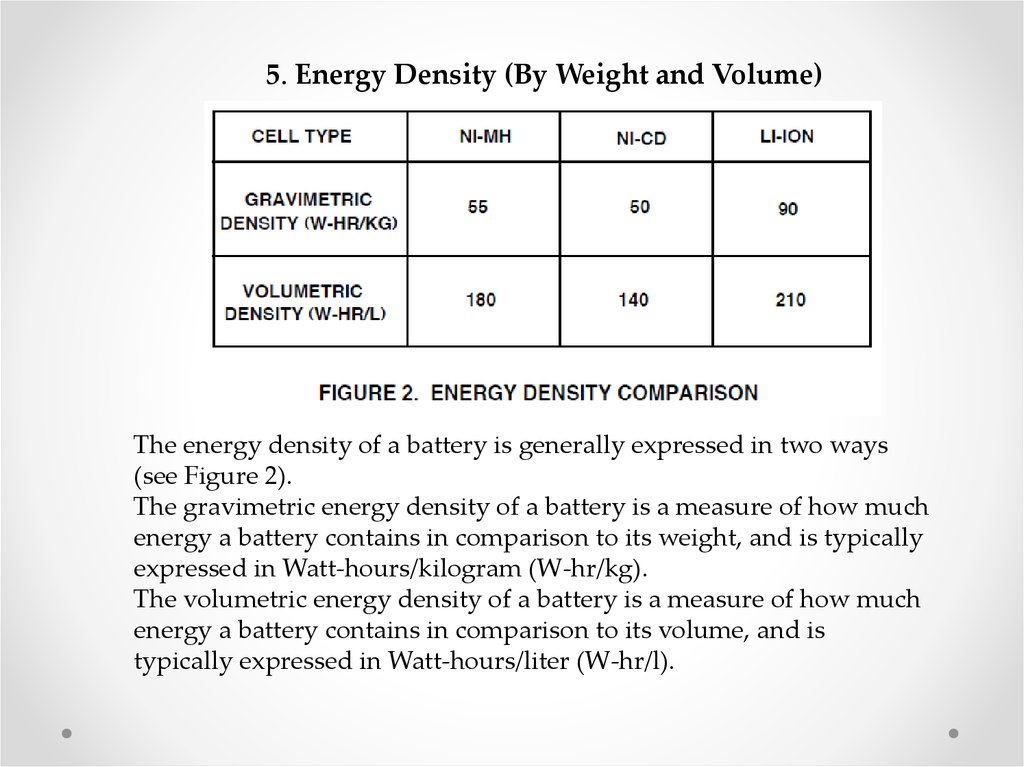
5. Energy Density (By Weight and Volume)The energy density of a battery is generally expressed in two ways
(see Figure 2).
The gravimetric energy density of a battery is a measure of how much
energy a battery contains in comparison to its weight, and is typically
expressed in Watt-hours/kilogram (W-hr/kg).
The volumetric energy density of a battery is a measure of how much
energy a battery contains in comparison to its volume, and is
typically expressed in Watt-hours/liter (W-hr/l).
8.
9.
6. Temperature dependenceThe rate of decrease of voltage with increasing discharge will also be
higher at lower temperatures, as will the capacity- this is illustrated by
the following graph:
10.
7. Service lifeBatteries can also be subjected to
premature death by:
Over-charging
Over-discharging
Short circuiting
Drawing more current than it
was designed to produce
extreme temperatures
physical shock or vibrations
Metallic dendrites
(battery death due to aging)
11.
8. Self DischargeSelf-discharge (which occurs in all batteries) determines the "shelf life" of
a battery.
In general, Li-Ion is the best of the lot, while Ni-Cd and Ni-MH are fairly
comparable to each other. Ni-Cd is typically a little better than Ni-MH,
but this may even out as Ni-MH manufacturing technology matures.










 physics
physics chemistry
chemistry








