Similar presentations:
Phase formation rules for high entropy alloys
1.
HIT 2008Phase Formation Rules for
High Entropy Alloys
Yong Zhang
University of Science and Technology Beijing
ICAMP5
2.
AcknowledgementsProf. GuoLiang Chen;
Prof. Hywel A Davies;
Prof. Peter K Liaw;
Prof. George Smith;
Prof. Zhaoping Lu;
XueFei Wang; YunJun Zhou;
FangJun Wang.
3.
OutlinesI. Background & Motivations
II. Results & Discussions
III. Summaries
4.
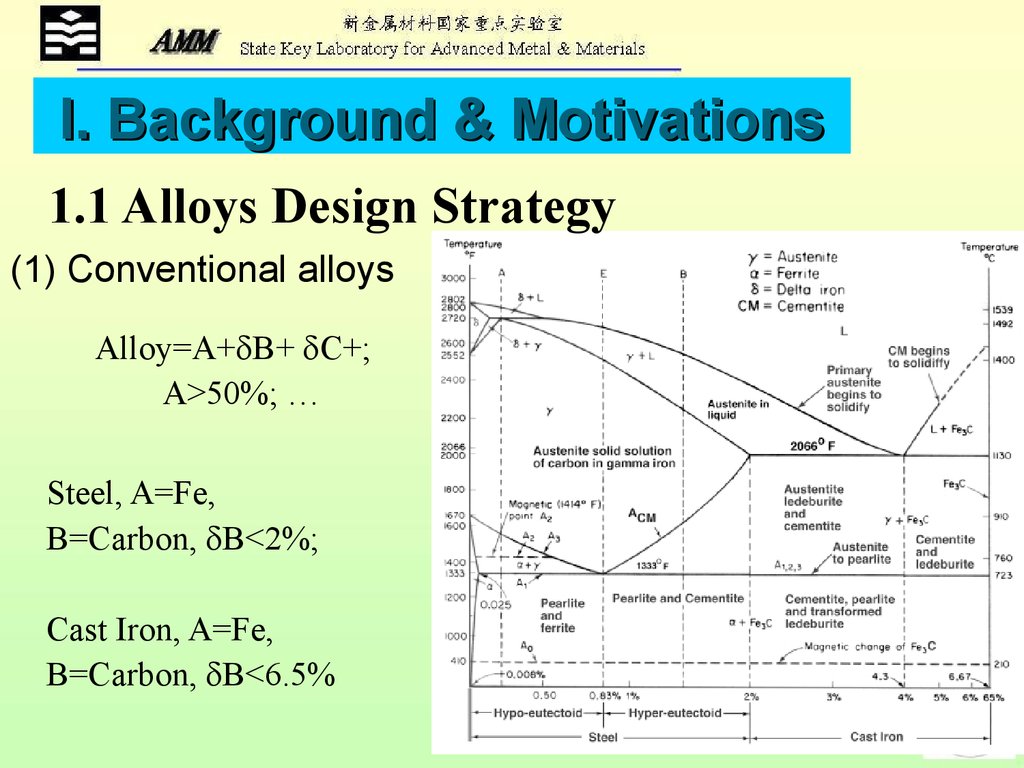
I. Background & Motivations1.1 Alloys Design Strategy
(1) Conventional alloys
Alloy=A+ B+ C+;
A>50%; …
Steel, A=Fe,
B=Carbon, B<2%;
Cast Iron, A=Fe,
B=Carbon, B<6.5%
5.
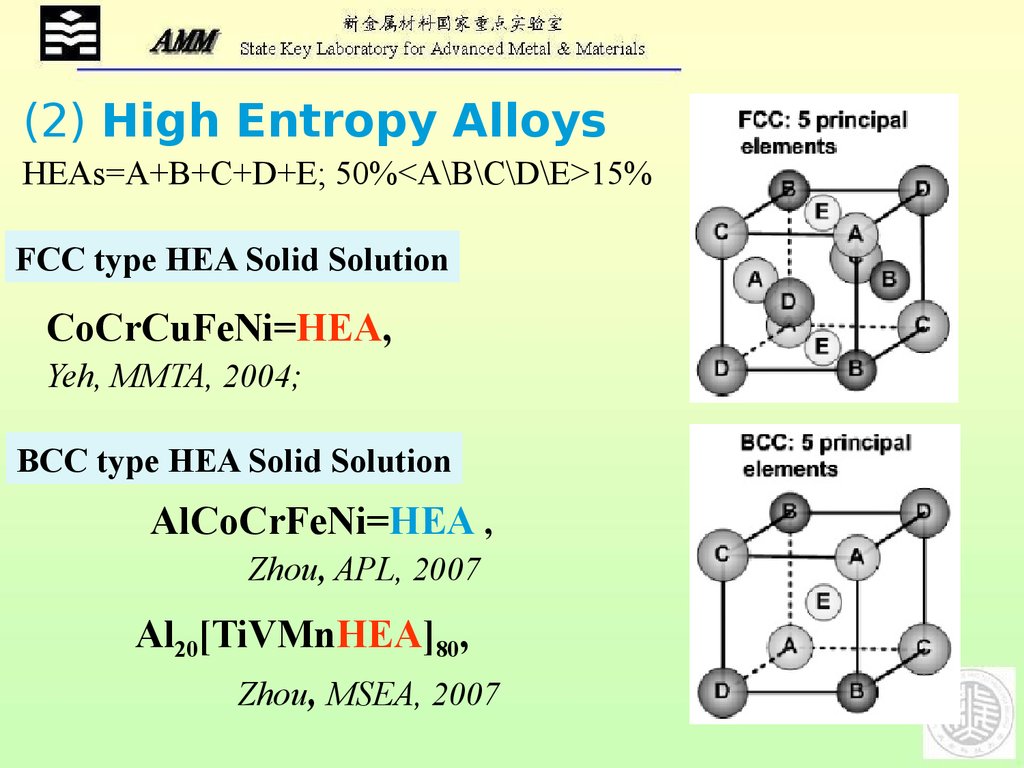
(2) High Entropy AlloysHEAs=A+B+C+D+E; 50%<A\B\C\D\E>15%
FCC type HEA Solid Solution
CoCrCuFeNi=HEA,
Yeh, MMTA, 2004;
BCC type HEA Solid Solution
AlCoCrFeNi=HEA ,
Zhou, APL, 2007
Al20[TiVMnHEA]80,
Zhou, MSEA, 2007
6.
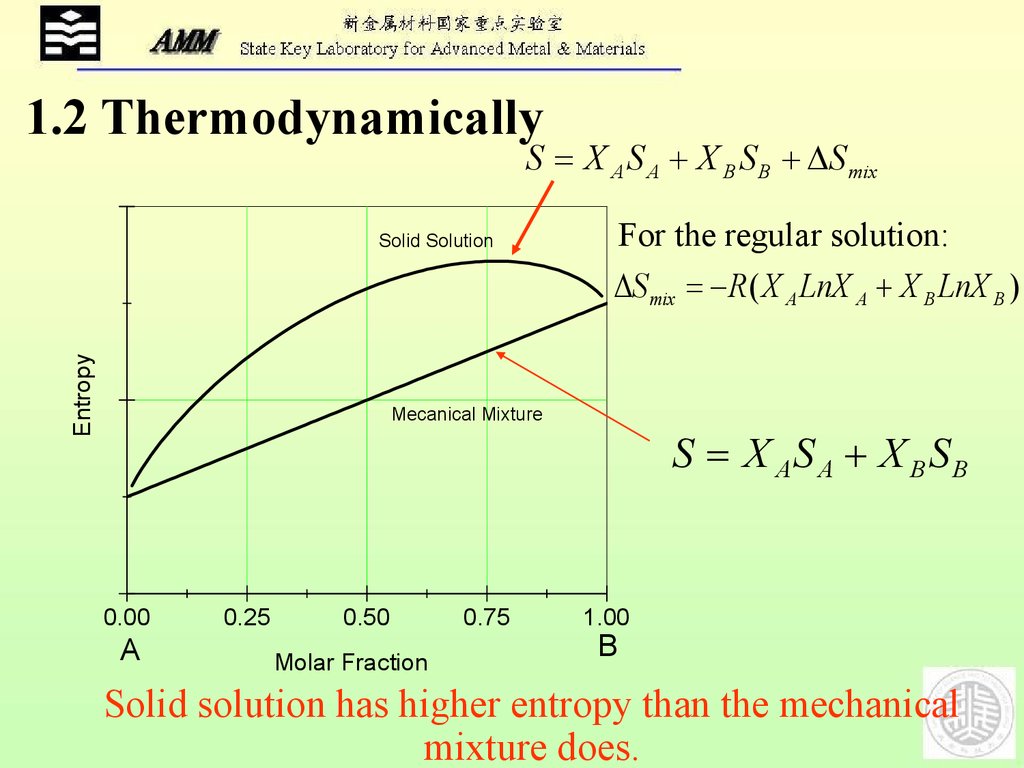
1.2 ThermodynamicallyS X A S A X B S B S mix
Entropy
Solid Solution
For the regular solution:
S mix R( X A LnX A X B LnX B )
Mecanical Mixture
0.00
A
0.25
0.50
Molar Fraction
0.75
S X AS A X B SB
1.00
B
Solid solution has higher entropy than the mechanical
mixture does.
7.
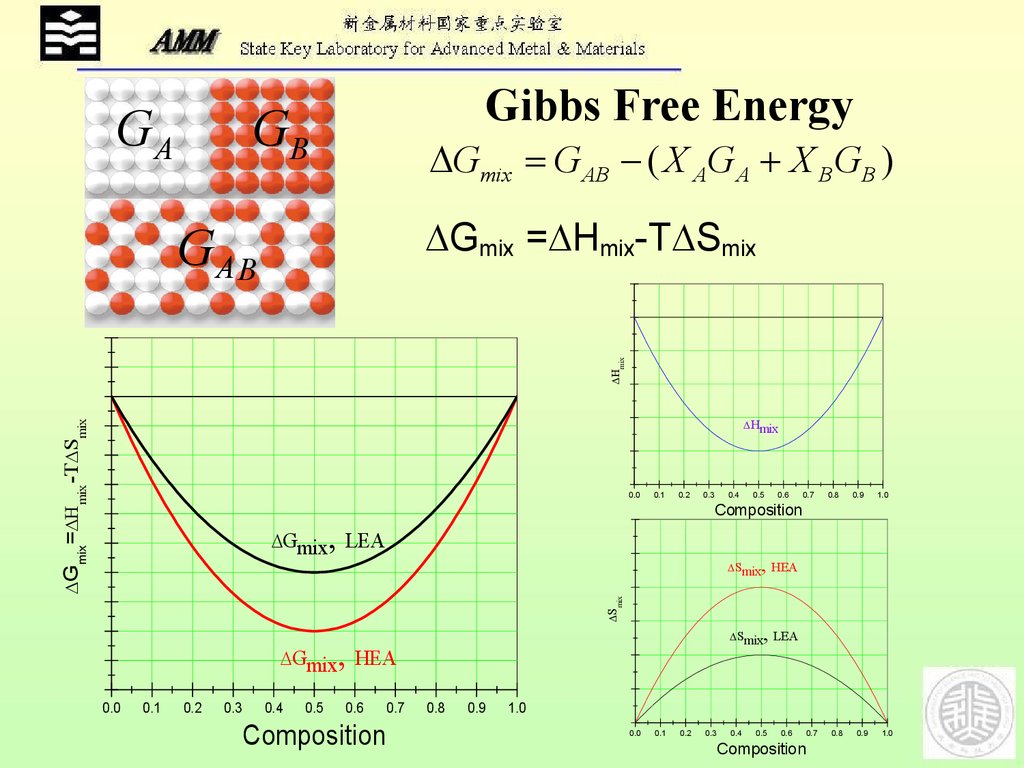
GAGibbs Free Energy
GB
Gmix G AB ( X AG A X B GB )
Gmix = Hmix-T Smix
mix
GA B
Gmix = mix -T Smix
Hmix
0.0
0.1
0.2
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1.0
Composition
,
Gmix LEA
,
Smix
Smix HEA
,
Smix LEA
,
Gmix HEA
0.0
0.1
0.2
0.3
0.4
0.5
0.6
0.7
Composition
0.8
0.9
1.0
0.0
0.1
0.2
0.3
0.4
0.5
0.6
Composition
0.7
0.8
0.9
1.0
8.
1.3 Properties and ApplicationsProperties
1. High Strength;
Zhou, APL, 2007;
2. High wear resistance; Lin, Surface
Coating technology, 2008.
3. High corrosion resistance; Lee, Thin
Solid Films, 2008;
4. High thermo-stability; Tsai, APL, 2008.
9.
Potential Applications1 Coatings, Barriers, etc.
Diffusion barriers for Cu interconnections; Tsai, APL, 2008
2 Structural Materials
3 Energy Storage Materials,
Raju, Journal of power Sources, 2008;
4 Molds
10.
1.4 MotivationsTo understand what is the dominant factors
for the phase formation of the HEAs
1 Atomic radius, or atomic volume;
The contents of Al, Ti, Cu, Co in
the HEAs were changed
N
c (1 r / r )
i 1
i
Atomic Radius
2
i
N
r Ci ri
i 1
Kittel, Introduction to Solid State Physics
11.
2 Enthalpy of Mixing;H mix
N
c c
i 1,i j
ij i
j
3 Entropy of Mixing
N
S mix R Ci LnCi
i 1
12.
4 Cooling RateCritical cooling rate? Like the BMG?
5 Tensile and compressive properties
Tensile elongation=0? Like BMG?
13.
II. Results & Discussions2.1. Alloying with different atomic size, Al, Cu, Co, Ti
Al=1.438A
(y=0, 0.25, 0.5, 0.75)
3.579A CoCrFeNiCu1-yAly
FCC
2.913A,2.872A
Ti0.5CoCrFeNiCu1-yAly
BCC, High APE to Lower APE, with larger atoms Al
14.
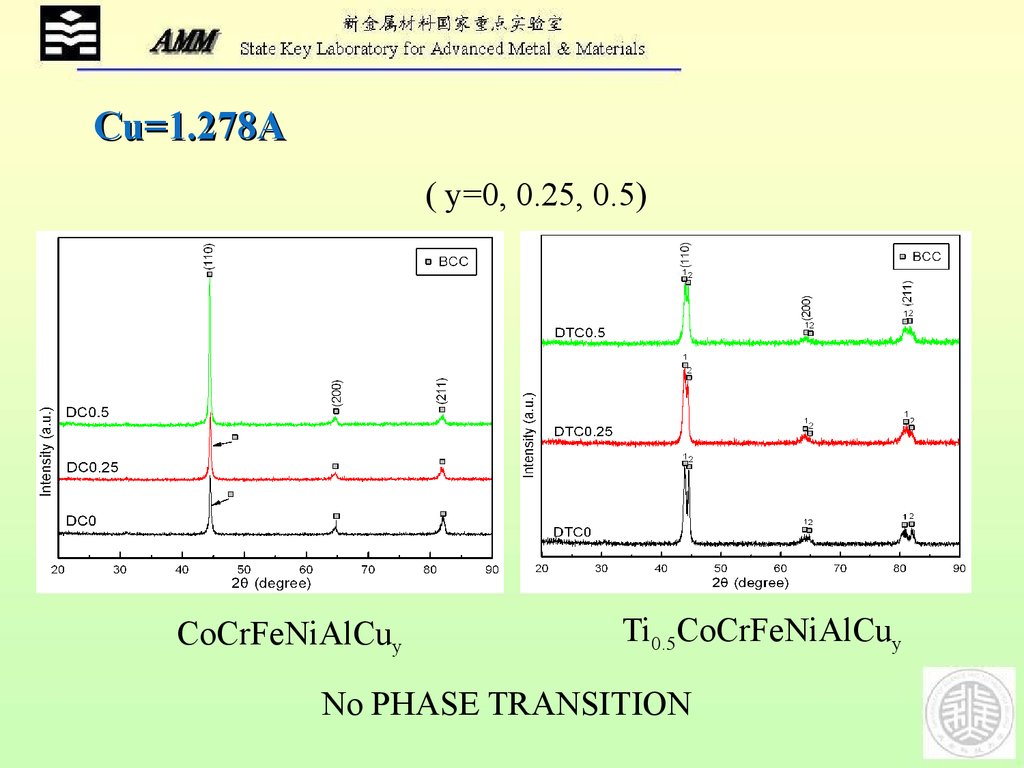
Cu=1.278A( y=0, 0.25, 0.5)
CoCrFeNiAlCuy
Ti0.5CoCrFeNiAlCuy
No PHASE TRANSITION
15.
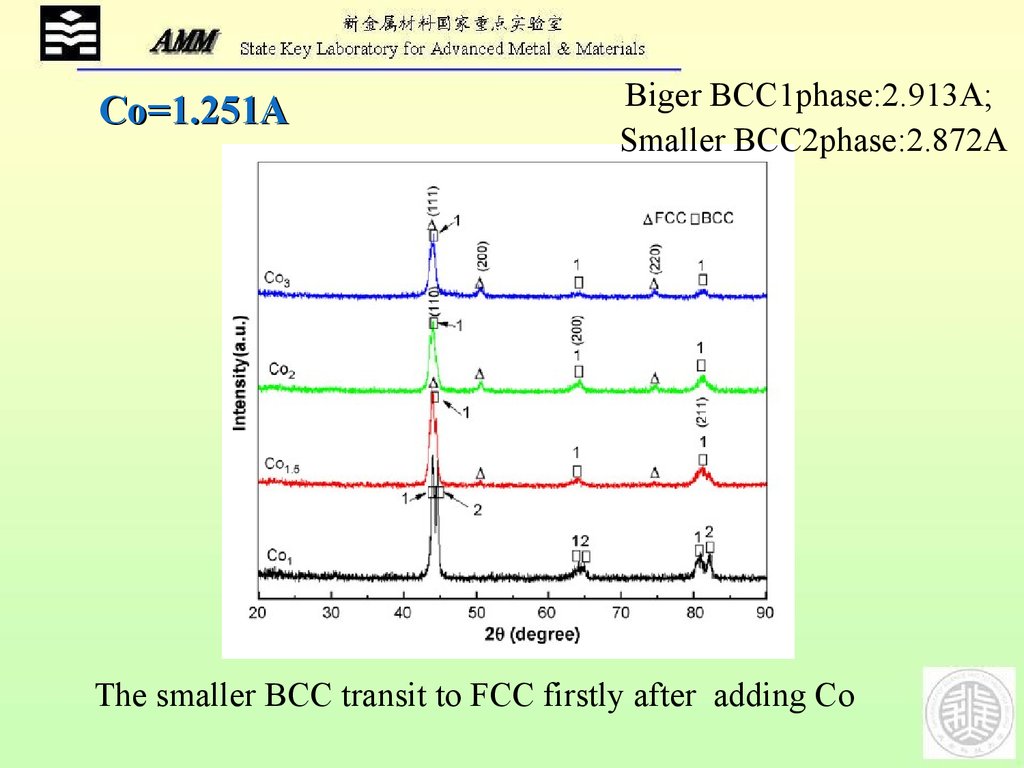
Co=1.251ABiger BCC1phase:2.913A;
Smaller BCC2phase:2.872A
The smaller BCC transit to FCC firstly after adding Co
16.
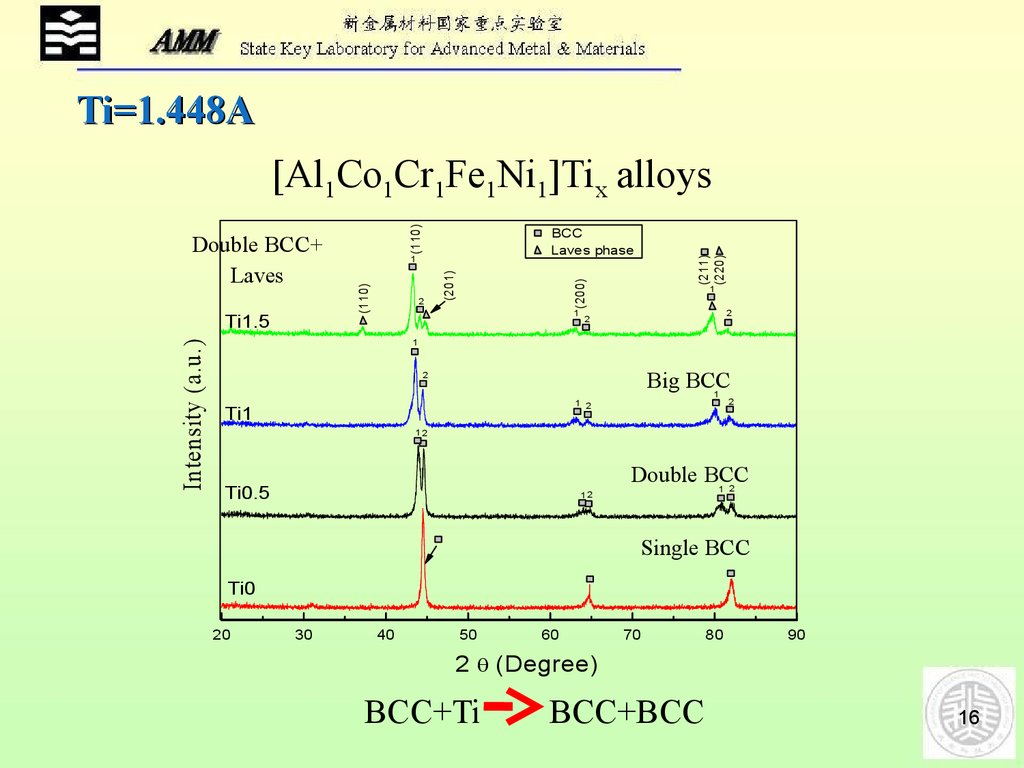
Ti=1.448AIntensity (a.u.)
Ti1.5
(200)
(201)
2
(211)
(220)
BCC
Laves phase
1
(110)
Double BCC+
Laves
(110)
[Al1Co1Cr1Fe1Ni1]Tix alloys
1
1
2
2
1
Big BCC
2
1
1 2
Ti1
2
12
Double BCC
Ti0.5
1 2
12
Single BCC
Ti0
20
30
40
50
60
70
80
90
2 (Degree)
BCC+Ti
BCC+BCC
16
17.
After adding Ti, Laves phase forms18.
The transition is mainly lattice distortion induced and APE relatedZhou, APL, 2008
19.
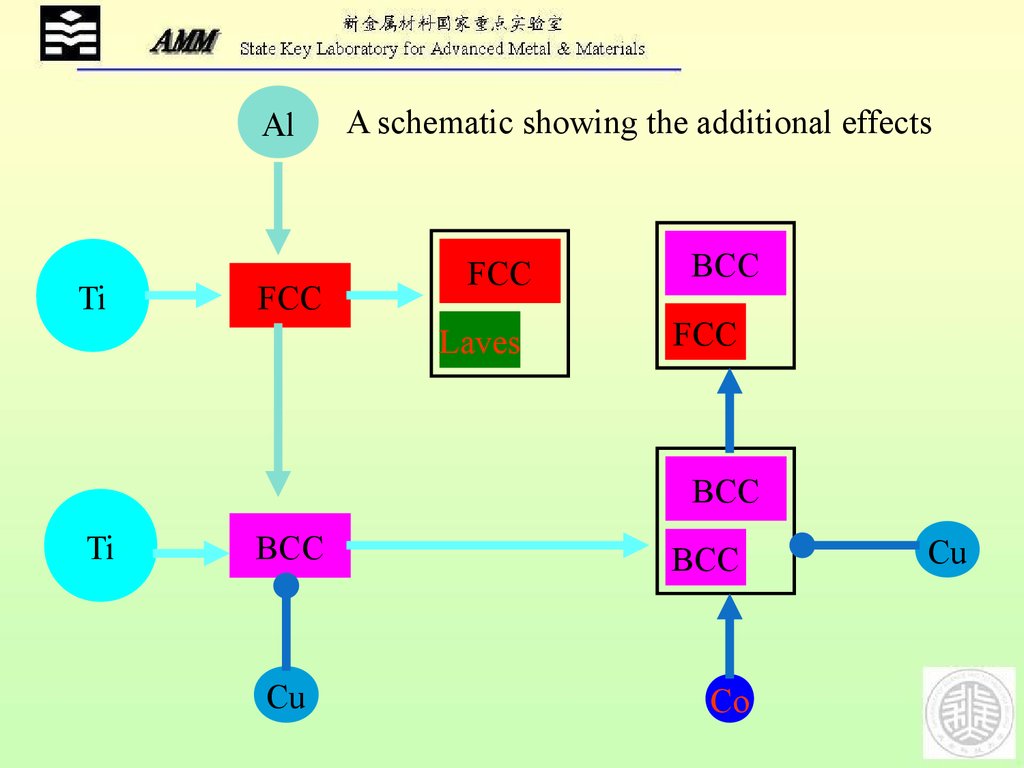
AlTi
FCC
A schematic showing the additional effects
FCC
Laves
BCC
FCC
BCC
Ti
BCC
Cu
BCC
Co
Cu
20.
2.2. Considering of the enthalpy of mixing HmixMg based BMG
Zr based BMG
20
Zhang, AEM, 2008
21.
2.3. Considering of the entropy of mixing SmixHigh Entropy is not good for the formation of BMG
21
22.
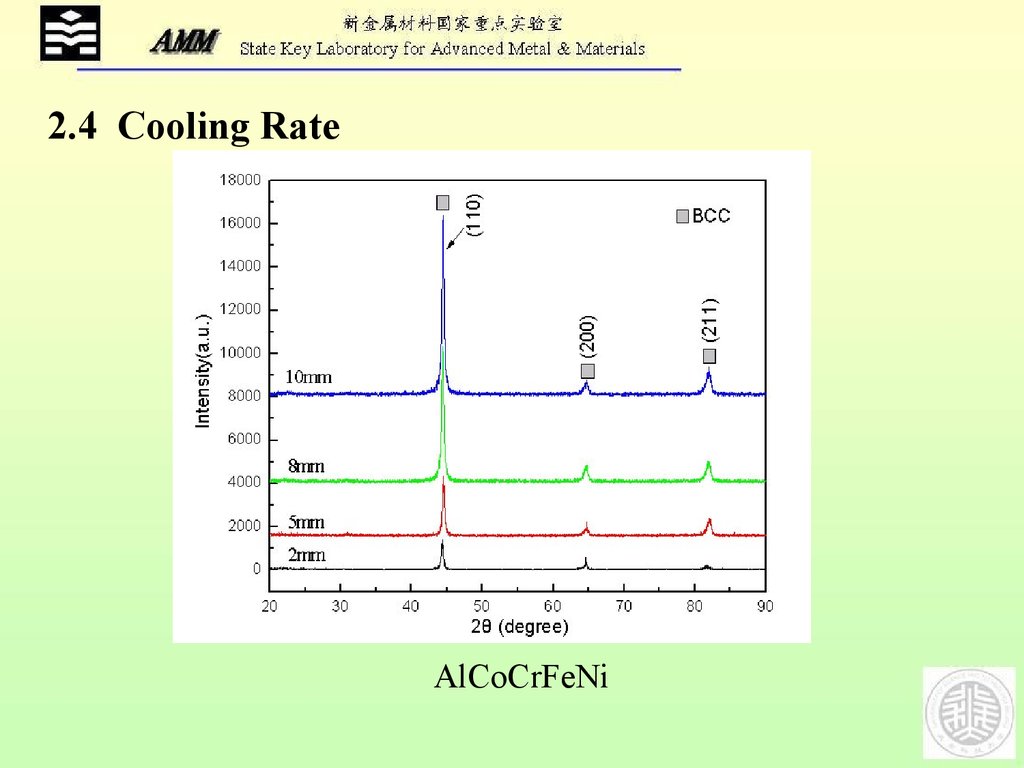
2.4 Cooling RateAlCoCrFeNi
23.
5mm2mm
8mm
AlCoCrFeNi
10mm
24.
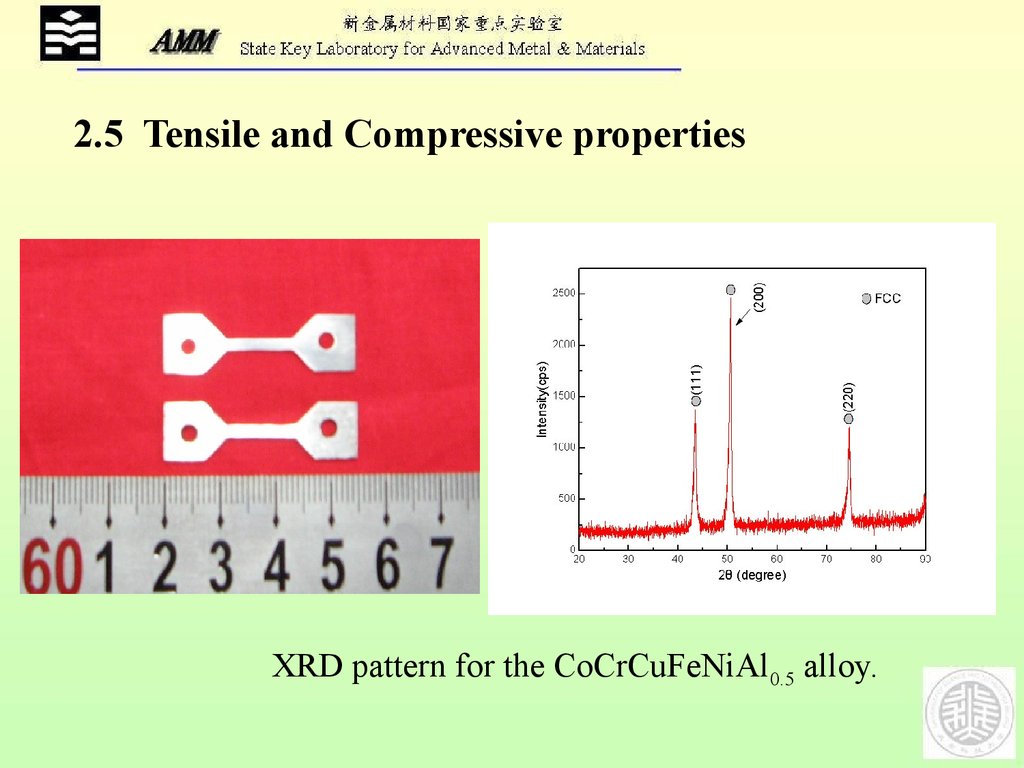
AlCoCrFeNi25.
2.5 Tensile and Compressive propertiesXRD pattern for the CoCrCuFeNiAl0.5 alloy.
26.
5 10Table Room temperature mechanical test results for the CoCrCuFeNiAl0.5 alloy
This alloy
P (%)
0.2 (MPa)
max (MPa)
Compressive
>51.5
460
>1380
Tensile
19.1
360
707
P: plastic strain; 0.2 : yield strength; max: compressive/tensile strength
27.
III. Summaries1 Atomic size mismatch is the dominant factor for the phase
formation of the high entropy alloys;
2 The formation of solid solution for the HEAs intends to have
enthalpy of mixing close to zero;
3 High entropy of mixing facilitates the formation of the solid
solution rather than the BMGs;
4 Cooling rate plays rather important role for the homogeneous
microstructure than for the phase formation;
5 HEA can have tensile elongations as high as 19%.
28.
Thanks for yourattention
28


















 physics
physics chemistry
chemistry








