Similar presentations:
The halogens
1.
1 of 37© Boardworks Ltd 2007
2.
2 of 37© Boardworks Ltd 2007
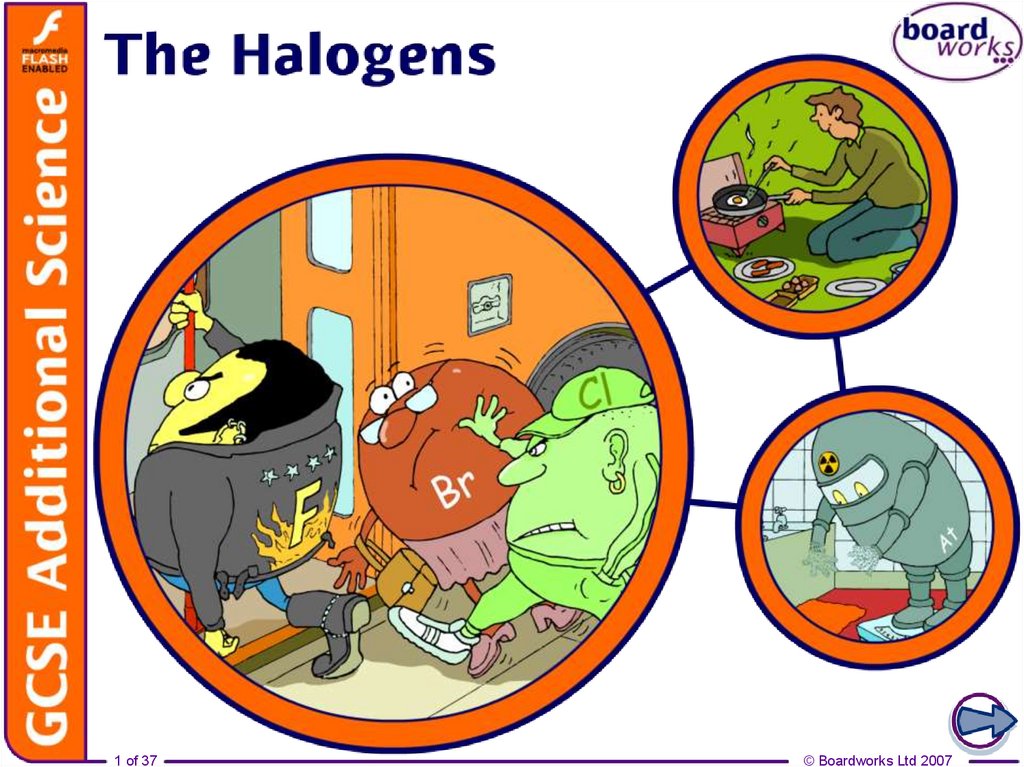
3. Group 7 – the halogens
The elements in group 7 of the periodic table, on the right,are called the halogens.
3 of 37
F
fluorine
Cl
chlorine
Br
bromine
I
iodine
At
astatine
© Boardworks Ltd 2007
4. Why are they called the ‘halogens’?
Halogens are very reactive non metals.They are all toxic or harmful
because they are so reactive.
Before antiseptics, iodine was
used to clean wounds as it is
harmful to all things, including
bacteria.
They are also never found free in nature because of their
reactivity – they are found as compounds with metals.
These halogen-metal compounds are salts, which give
halogens their name – ‘halo-gen’ means ‘salt-former’.
4 of 37
© Boardworks Ltd 2007
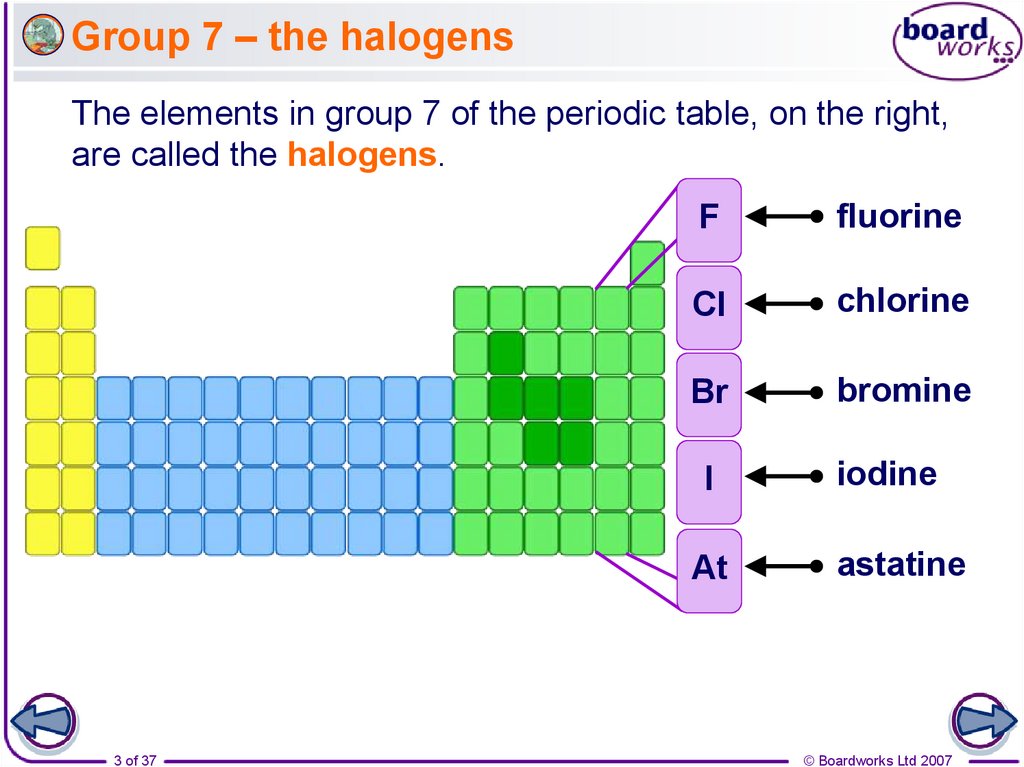
5. What is the electron structure of the halogens?
All halogens have seven electrons in their outer shell.This means that:
fluorine
They can easily obtain a
2,7
full outer shell by gaining
one electron.
They all gain an electron
in reactions to form
negative ions with a -1
charge.
They have similar
chemical properties.
5 of 37
chlorine
2,8,7
bromine
2,8,8,7
© Boardworks Ltd 2007
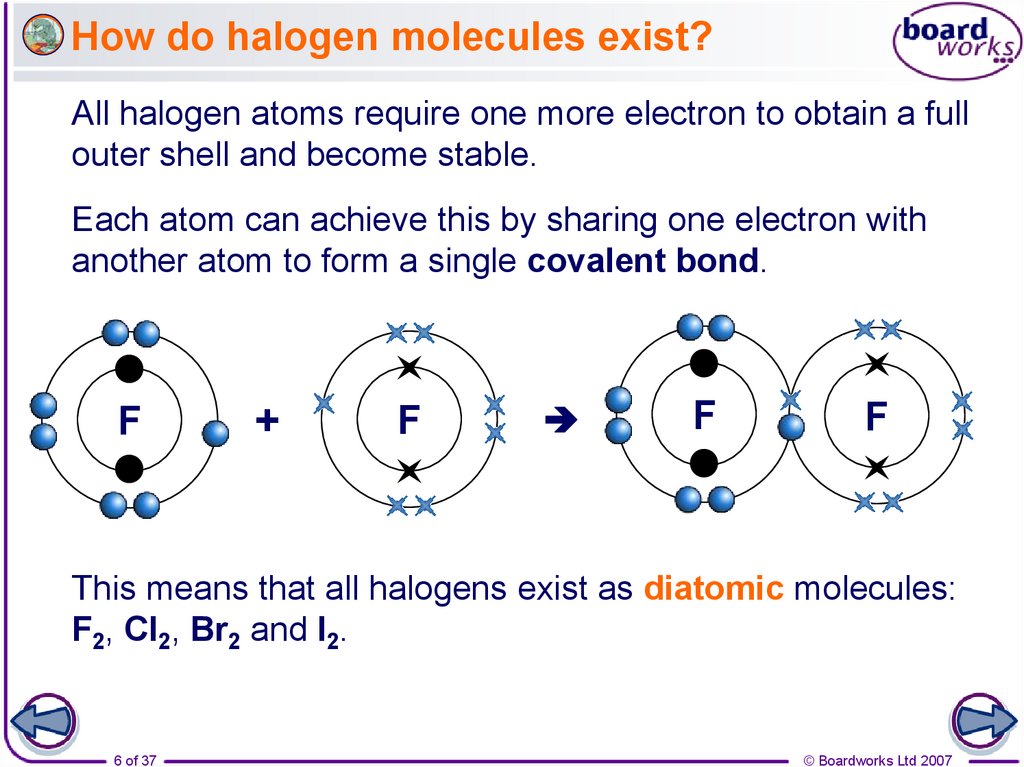
6. How do halogen molecules exist?
All halogen atoms require one more electron to obtain a fullouter shell and become stable.
Each atom can achieve this by sharing one electron with
another atom to form a single covalent bond.
F
+
F
F
F
This means that all halogens exist as diatomic molecules:
F2, Cl2, Br2 and I2.
6 of 37
© Boardworks Ltd 2007
7.
7 of 37© Boardworks Ltd 2007
8. What are the general properties of the halogens?
All the halogens are:non-metals and so do not conduct electricity
brittle and crumbly when solid
poisonous and smelly.
They become darker in colour down the group:
is pale yellow
is green-yellow
is red-brown
is blue-black.
8 of 37
© Boardworks Ltd 2007
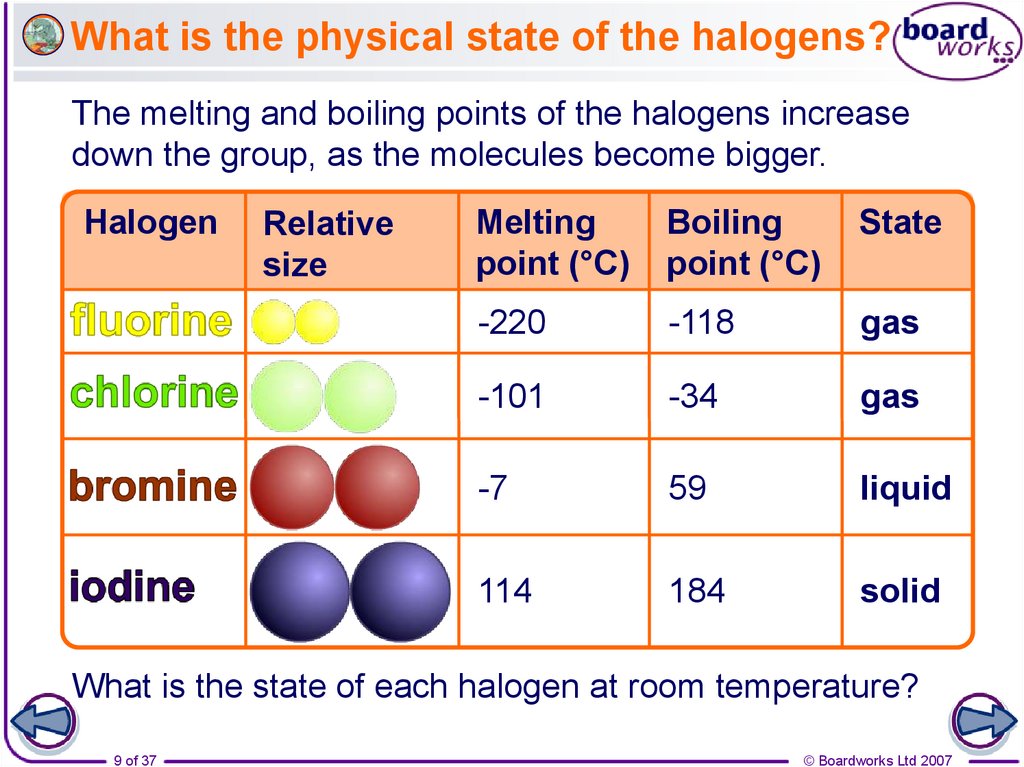
9. What is the physical state of the halogens?
The melting and boiling points of the halogens increasedown the group, as the molecules become bigger.
Halogen
Relative
size
Melting
point (°C)
Boiling
point (°C)
State
-220
-118
gas
-101
-34
gas
-7
59
liquid
114
184
solid
What is the state of each halogen at room temperature?
9 of 37
© Boardworks Ltd 2007
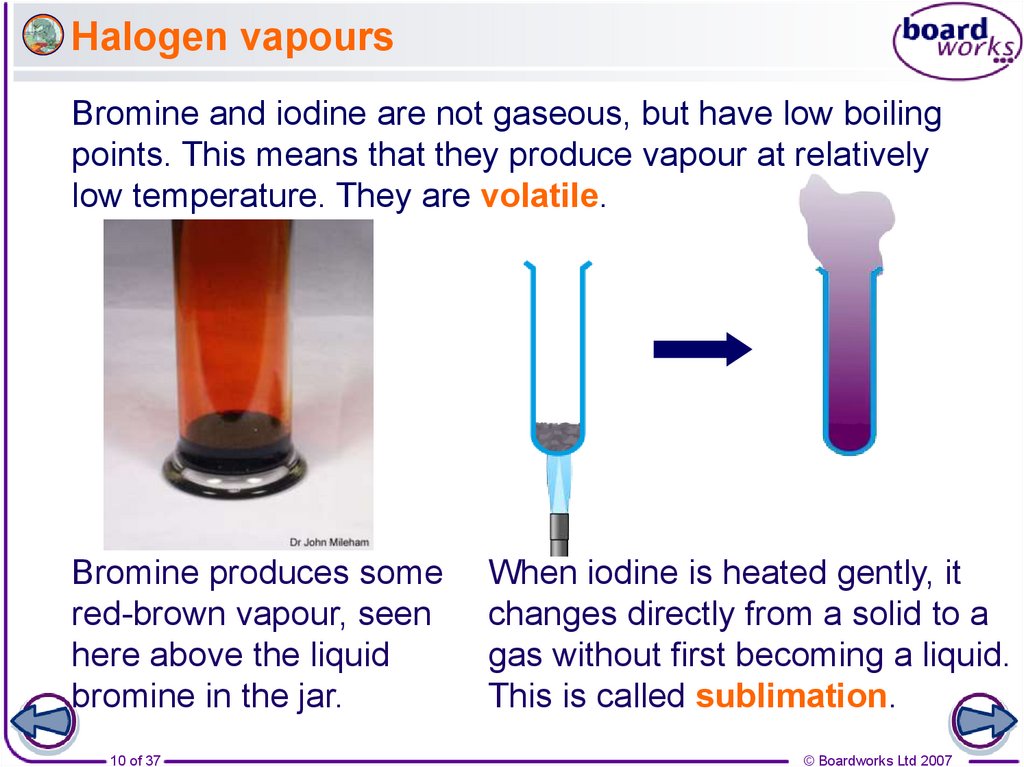
10. Halogen vapours
Bromine and iodine are not gaseous, but have low boilingpoints. This means that they produce vapour at relatively
low temperature. They are volatile.
Bromine produces some
red-brown vapour, seen
here above the liquid
bromine in the jar.
10 of 37
When iodine is heated gently, it
changes directly from a solid to a
gas without first becoming a liquid.
This is called sublimation.
© Boardworks Ltd 2007
11.
11 of 37© Boardworks Ltd 2007
12. How do the halogens react with metals?
The reactivity of halogens means that they readily react withmost metals.
Halogens need to gain electrons for a full electron shell and
metals need to lose electrons for a full electron shell.
This means that halogens and metals react to form ionic
compounds.
These are metal halides, which are a type of salt.
nickel (II) chloride
12 of 37
copper (II) chloride
© Boardworks Ltd 2007
13. What are halides?
When halogens react with another substance, they becomenegative ions, as they are gaining an extra electron.
When this happens, they are called halides.
The name of each of the halogens changes slightly once
it has reacted – instead of ending with ‘–ine’, they end
with ‘-ide’.
Halogen
13 of 37
reaction
Halide
(F)
fluoride (F-)
(Cl)
chloride (Cl-)
(Br)
bromide (Br-)
(I)
iodide (I-)
© Boardworks Ltd 2007
14. Equations of halogens and iron
When a halogen reacts with iron it forms an iron halide:halogen
+
iron
iron (III) halide
The word and chemical equations for the reaction between
chlorine and iron are:
iron (III)
chlorine + iron
chloride
3Cl2 (g)
+ 2Fe (s)
2FeCl3 (s)
What would the equation be for the reaction that forms
iron (III) bromide?
14 of 37
bromine
+ iron
iron (III)
bromide
3Br2 (g)
+ 2Fe (s)
2FeBr3 (s)
© Boardworks Ltd 2007
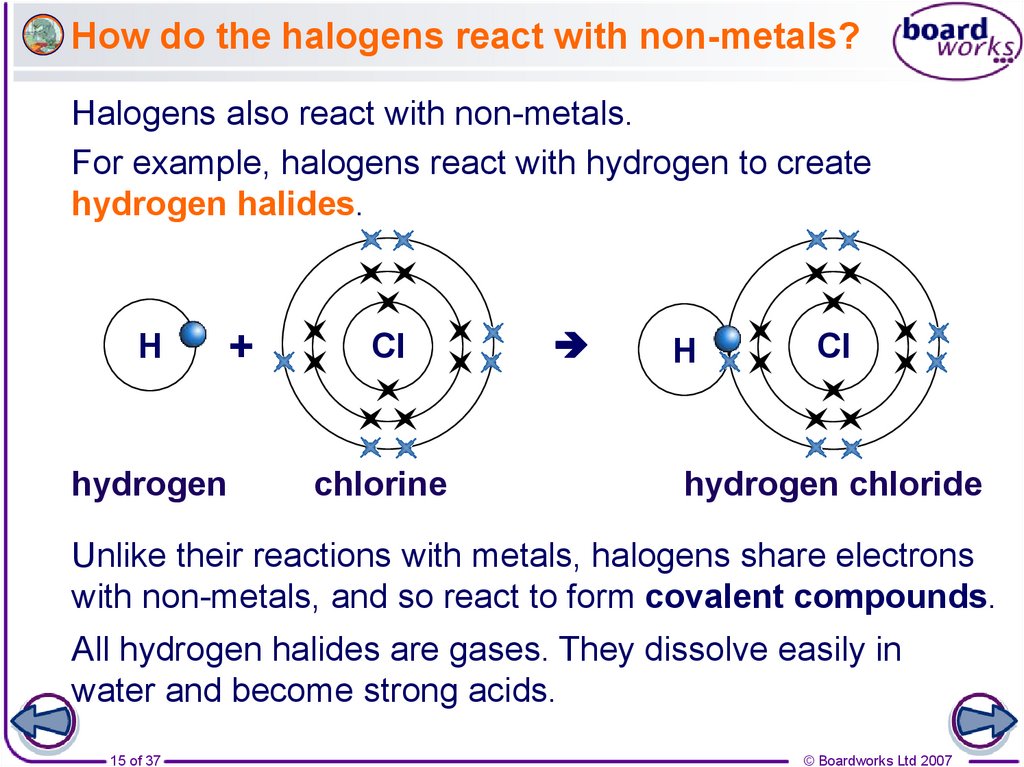
15. How do the halogens react with non-metals?
Halogens also react with non-metals.For example, halogens react with hydrogen to create
hydrogen halides.
H
hydrogen
+
Cl
chlorine
H
Cl
hydrogen chloride
Unlike their reactions with metals, halogens share electrons
with non-metals, and so react to form covalent compounds.
All hydrogen halides are gases. They dissolve easily in
water and become strong acids.
15 of 37
© Boardworks Ltd 2007
16. Displacement of halogens
If a halogen is added to a solution of a compound containinga less reactive halogen, it will react with the compound and
form a new one.
This is called displacement.
fluorine
sodium
chloride
+
sodium
fluoride
+
chlorine
F2 (aq)
+ 2NaCl (aq)
2NaF (aq)
+
Cl2 (aq)
A more reactive halogen will always displace a less reactive
halide from its compounds in solution.
16 of 37
© Boardworks Ltd 2007
17. Displacement of halogens
Why will a halogen always displace a less reactive halogen?17 of 37
© Boardworks Ltd 2007
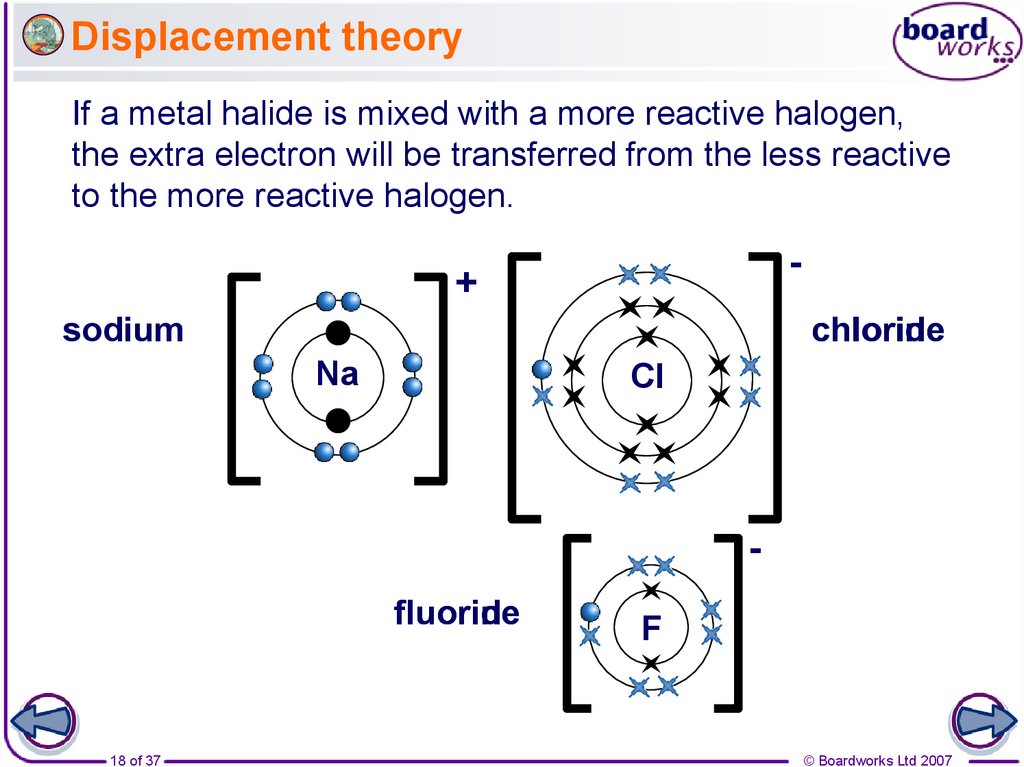
18. Displacement theory
If a metal halide is mixed with a more reactive halogen,the extra electron will be transferred from the less reactive
to the more reactive halogen.
--
+
chlorine
chloride
sodium
Na
Cl
fluorine
fluoride
18 of 37
F
© Boardworks Ltd 2007
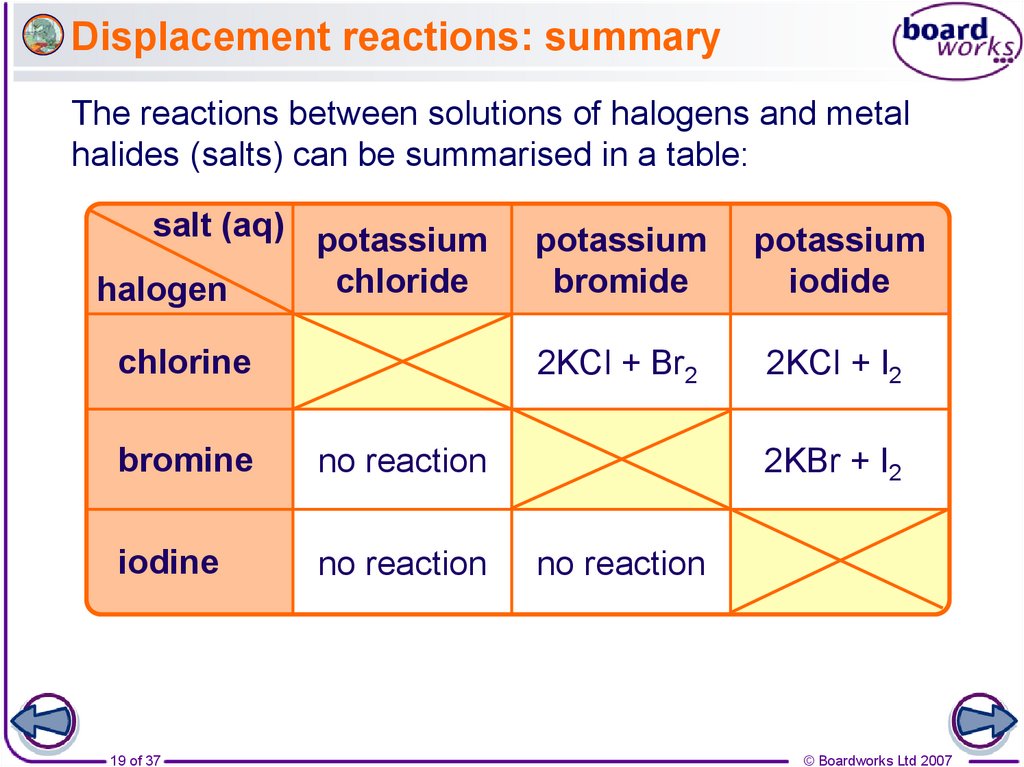
19. Displacement reactions: summary
The reactions between solutions of halogens and metalhalides (salts) can be summarised in a table:
salt (aq) potassium
chloride
halogen
potassium
bromide
potassium
iodide
chlorine
2KCl + Br2
2KCl + I2
bromine
no reaction
iodine
no reaction
19 of 37
2KBr + I2
no reaction
© Boardworks Ltd 2007
20.
20 of 37© Boardworks Ltd 2007
21. What are the uses of halogens?
21 of 37© Boardworks Ltd 2007
22.
22 of 37© Boardworks Ltd 2007













 chemistry
chemistry